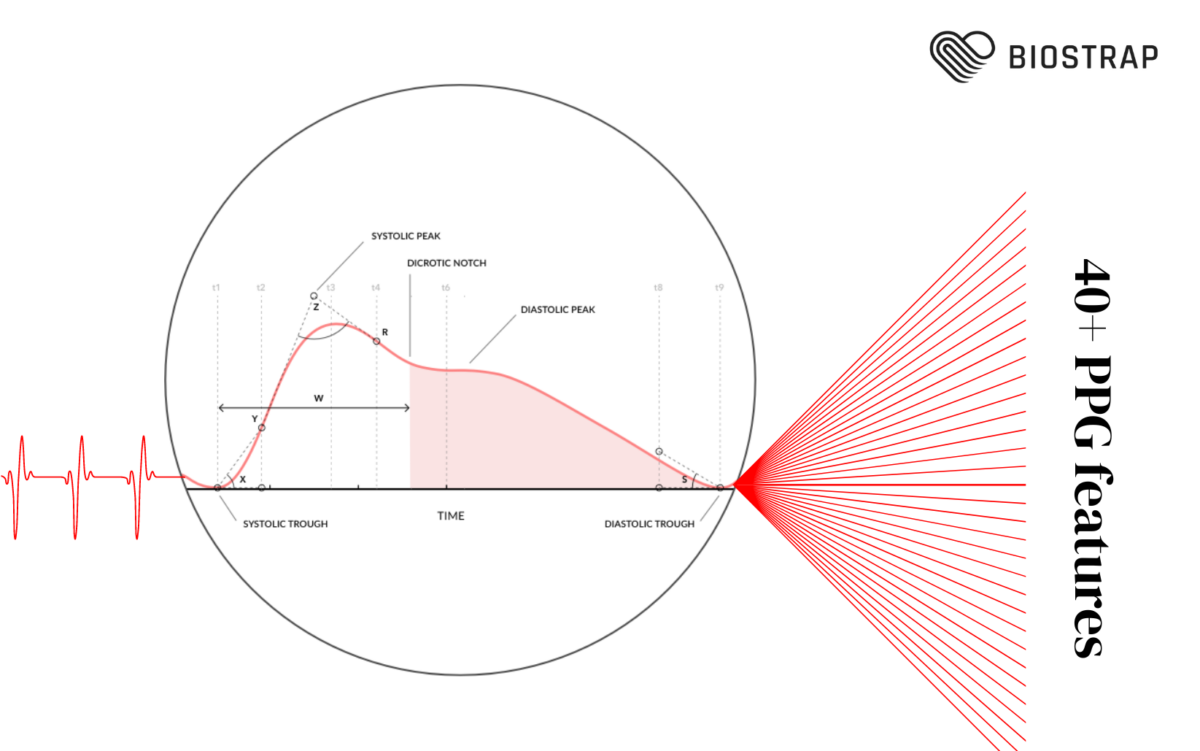
Ever feel like your body’s sending you mixed signals? One moment you’re wound up tighter than a spring, and the next, you’re too drained to move. It’s like being stuck in traffic with your emotions honking loudly, trying to signal you through the fog. Understanding whether you’re dealing with emotional or physical stress isn’t just about putting a name to your pain; it’s about finding the right tools in your self-care kit to deal with it. Distinguishing between these two types of stress is crucial in order to manage them effectively.
Emotional or physical stress
Picture this: Emotional stress is like having a bunch of browser tabs open in your brain—constantly loading, never quite settling. It stems from worries, anxieties, and the mental burdens we carry. Physical stress, on the other hand, is the body’s way of saying, “Hey, remember that high-intensity interval training session at the gym? I’m still dealing with that.”
Why does the distinction matter? Because the remedy for a mind cluttered with worries is different from the cure for muscles aching from overexertion. Recognizing the source of your stress is step one on the path to managing it.
How to tell them apart
Feeling stuck in a mental loop, ruminating over past conversations or future anxieties? That’s your cue for emotional stress. On the flip side, if your body feels like it’s been through a wringer after a physically demanding day, you’re looking at physical stress. The key to differentiation lies in self-awareness—listening to what your body and mind are telling you. Here’re are six actionable tips for managing stress – both physical and emotional.
Caught in a thought tornado? Go for a walk.
When your mind is a maze of worries, a change of scenery can work wonders. Walking isn’t just good for your body; it’s a balm for the mind, too. It’s about hitting the refresh button on your thoughts and letting nature’s calm infiltrate your mental chaos.
Feeling down? Hit the gym.
If emotional stress has you feeling low, exercise can be your elevator. It’s not just about staying fit; it’s about releasing endorphins, those feel-good hormones that act like natural painkillers and mood lifters. Whether it’s a dance class, a quick jog, or yoga, moving your body can help clear the emotional clutter.
In a Funk? Get some sun.
Never underestimate the power of a sunny day. Sunlight is nature’s mood enhancer, boosting your serotonin levels and battling the blues. So, if you’re feeling down, step outside and let the sun’s rays lift your spirits. Even a brief lunch break under the sky can make a difference.
Muscles screaming after a workout? Prioritize recovery.
If physical stress has left your muscles shouting objections, it’s time to listen. Rest is not laziness; it’s an essential part of the recovery process. Combine it with strategies like a warm bath, sauna (if you can), gentle stretching, or massage, and you’re not just treating your body; you’re thanking it.
Overwhelmed by emotions? Write It down.
Sometimes, the act of transferring your thoughts from mind to paper can help untangle them. Journaling is a therapeutic way to face emotional stress head-on. It’s about giving your feelings a place to rest outside of your head.
Breathe through it: The power of mindful breathing.
Whether it’s emotional anxiety or the aftermath of a physical strain, mindful breathing can be a bridge to calm. Deep, deliberate breaths signal your body that it’s safe to relax, turning down the volume on stress.
Why it all matters
Understanding the nature of your stress is like having the right key for the lock. It empowers you to choose the most effective strategies for relief, ensuring that you’re not just throwing solutions at the problem and hoping something sticks. It’s about targeted, effective self-care that acknowledges the complexity of human experience—recognizing that we are both mind and body, intertwined.
Deciphering whether your stress is emotional or physical is more than a mental exercise; it’s a critical step toward holistic well-being. By tuning into the signals your body and mind are sending, you can tailor your self-care practices to meet your needs effectively. Whether it’s through movement, mindfulness, or simply soaking up some sun, the path to managing stress starts with understanding its roots. Remember, in the journey toward wellness, every step, every breath, and every moment of mindfulness counts.